
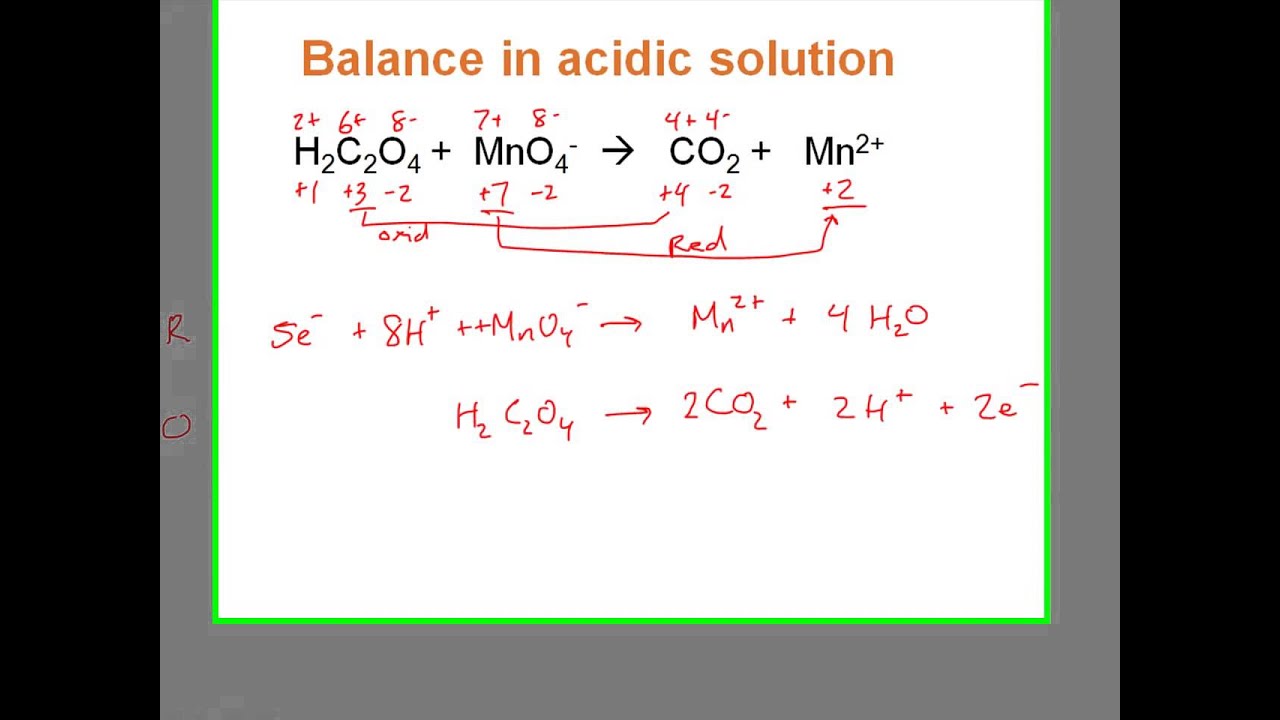
Notice that, in the answer, the S coefficient stays the same (but the subscript of 8 goes away) and the other coefficients are all reduced by a factor of 8. Also, note that duplicates of 48 electrons and 48 hydrogen ions were removed.ĥ) Sometimes, you will see the nitric acid in molecular form:Ģ4H 2S + 16HNO 3 -> 3S 8 + 16NO + 32H 2OĮxample #2b: H 2S + HNO 3 -> NO + S + H 2Oĭiscussion: Many times, teachers and textbooks will use S rather than S 8. The duplicates are 6e¯, 3H 2O, and 6H +Ģ4H 2S -> 3S 8 + 48H + + 48e¯ 16NO + 32H 2O 3S 8 + 16NO + 32H 2OĬomment: removing a factor of 8 does look tempting, doesn't it? However, the three in front of the S 8 (or the five in the next example) makes it impossible. Note that items duplicated on each side were cancelled out. These items are usually the electrons, water and hydrogen ion.Īfter Example #5c, I have a suggestion for searching for help on balancing a specific equation.Įxample #1: ClO 3¯ + SO 2 -> SO 4 2¯ + Cl¯ĦH 2O + 3SO 2 -> 3SO 4 2¯ + 12H + + 6e¯ 3SO 4 2¯ + Cl¯ + 6H + In order to get the electrons in each half-reaction equal, one or both of the balanced half-reactions will be multiplied by a factor.Ģ) Duplicate items are always removed. Multiply the number of calcium atoms by 3 and the number of phosphorus atoms by 2.Balancing redox reactions in acidic solution Balancing redox reactions in acidic solutionįifteen Examples Problems 1-10 Problems 26-50 Balancing in basic solution Problems 11-25 Only the examples and problems Return to Redox menuġ) Electrons NEVER appear in a correct, final answer. Phosphorus goes from 0 to -3, gaining 3 electrons (oxidation). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
